Chemistry (0620) Experimental Techniques and Chemical Analysis Revision Note
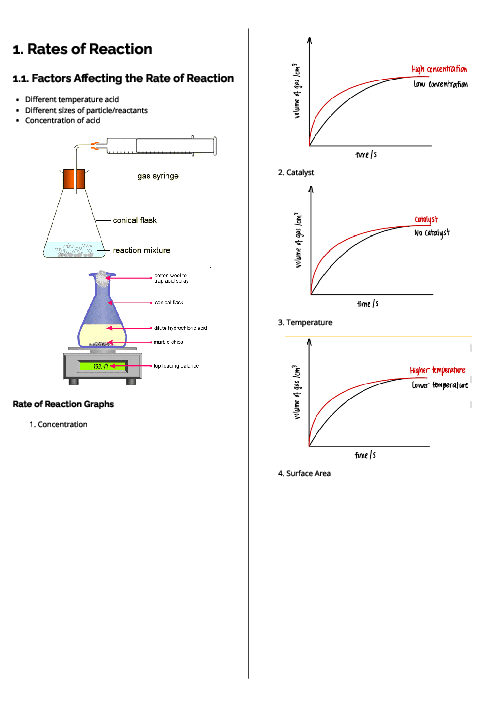
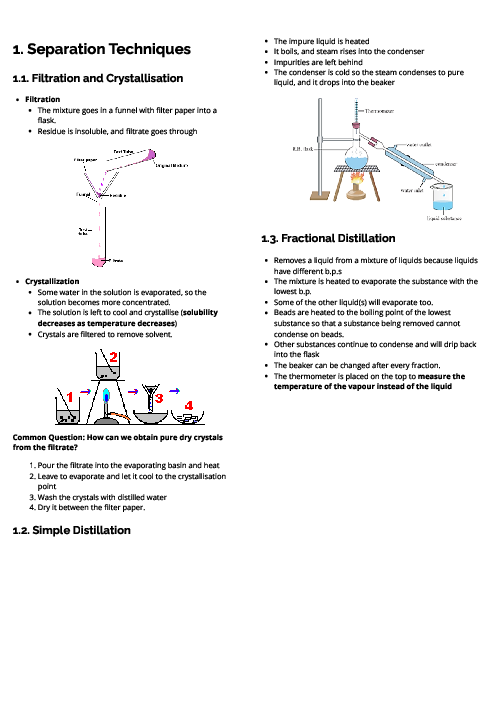
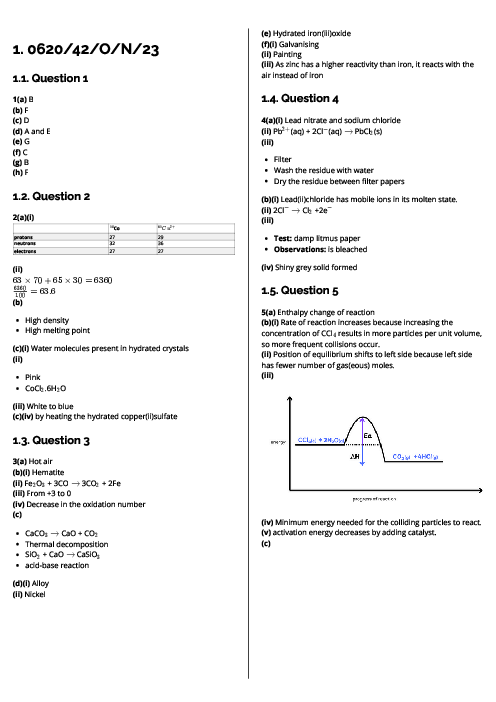
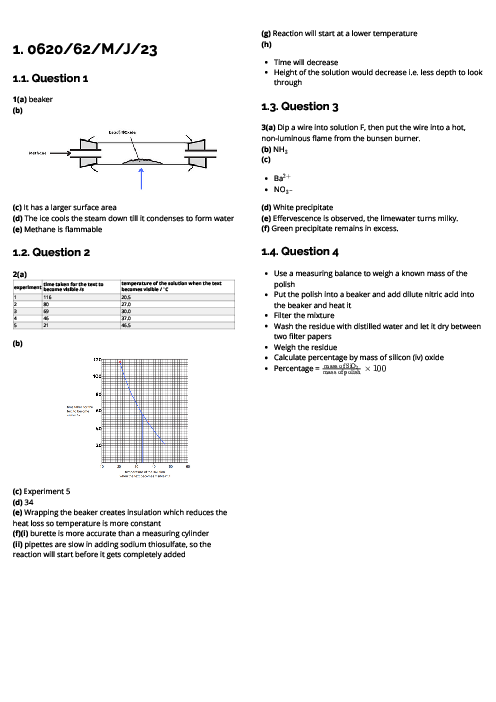
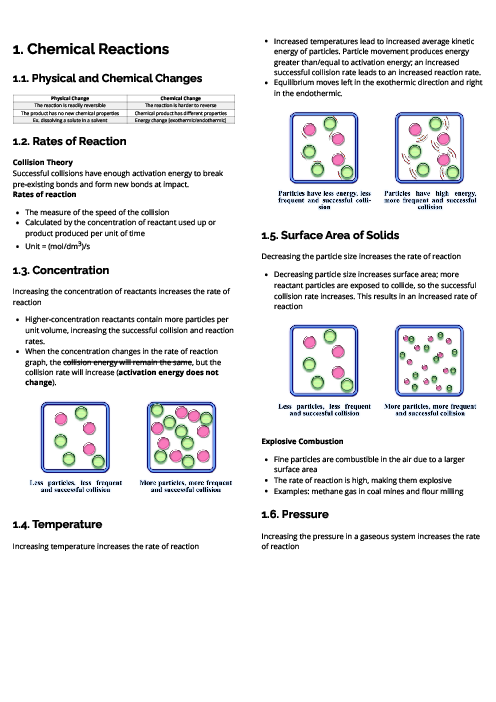
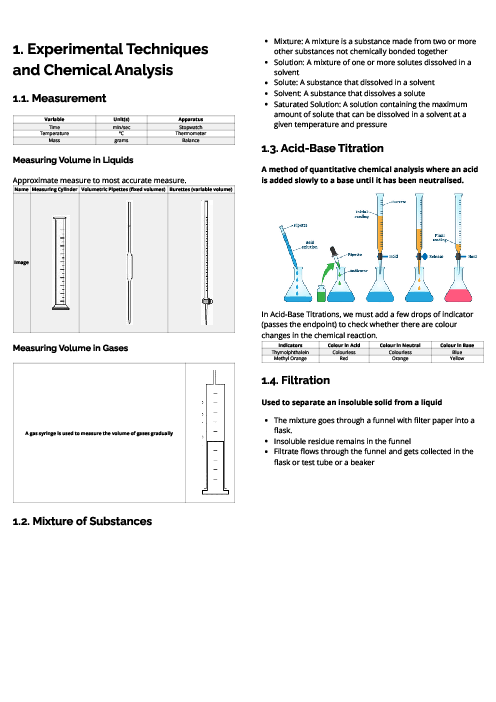
This document explains experimental techniques and chemical analysis for CAIE IGCSE Chemistry, covering measurement, separation methods, purification, and tests for ions and gases. It begins with measurement techniques, including the use of stopwatches, thermometers, balances, pipettes, burettes, and gas syringes. Methods of separating mixtures are detailed: filtration for insoluble solids, crystallisation for dissolved solids, simple distillation for solvents, fractional distillation for miscible liquids, and chromatography for separating substances based on solubility and retention factor (Rf value). Practical techniques like acid-base titration are explained with indicators such as thymolphthalein and methyl orange. The section on purity highlights that pure substances have sharp melting/boiling points, while impurities alter these values. The guide provides detailed tests for cations (e.g., aluminium, ammonium, calcium, copper, iron, zinc, chromium), anions (carbonate, chloride, bromide, iodide, nitrate, sulfate, sulfite), and gases (ammonia, CO₂, chlorine, hydrogen, oxygen, sulfur dioxide). Flame tests for metal ions are included: lithium (red), sodium (yellow), calcium (orange-red), potassium (lilac), barium (light green), and copper (blue-green). These tests are essential for qualitative analysis in exams. By mastering these experimental techniques, students can confidently approach both practical and theoretical components of chemistry assessments.
باز نشر محتواها در فضای مجازی، ممنوع است.
باز نشر محتواها در فضای مجازی، ممنوع است.