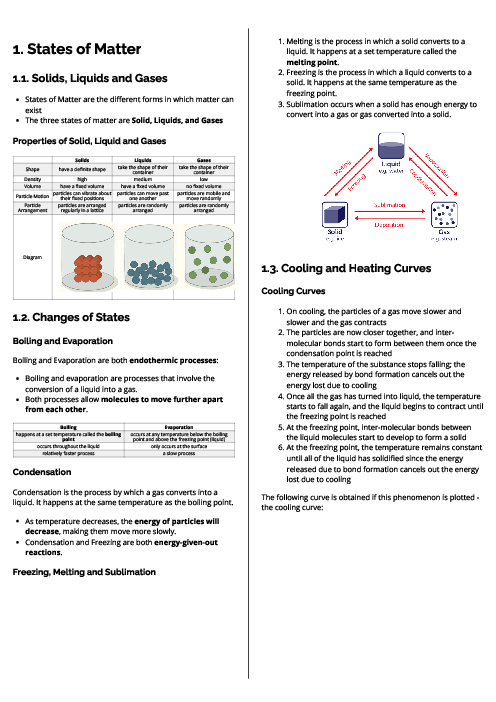
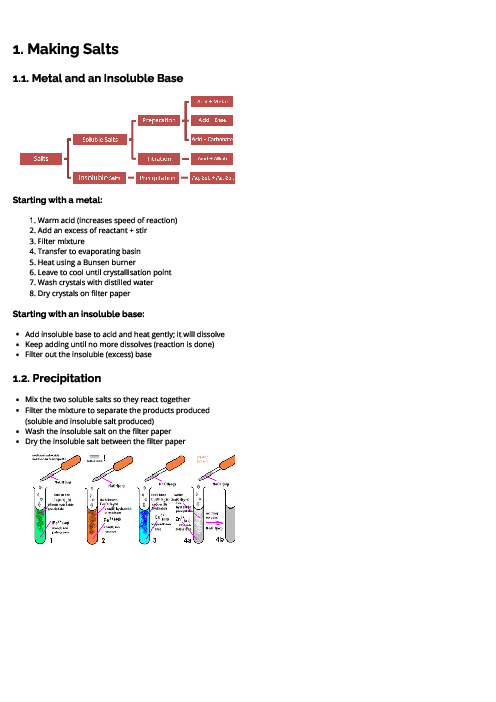
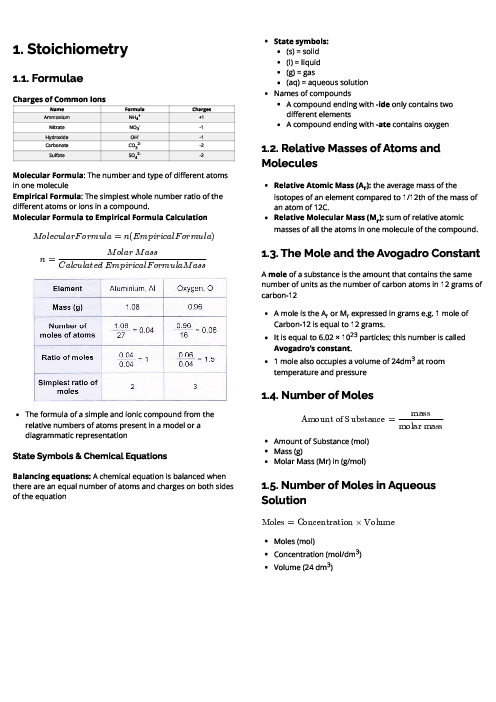
Chemistry (0620) Chemical Reactions Revision Note
This document explains chemical reactions for CAIE IGCSE Chemistry, focusing on reaction types, rates, equilibrium, and industrial processes. It begins with the difference between physical and chemical changes—physical changes are reversible with no new substances, while chemical changes form products with new properties and energy transfer. The rates of reaction are explained using collision theory, showing how concentration, temperature, surface area, and pressure affect the frequency of successful collisions. Catalysts and enzymes are introduced as substances that lower activation energy and speed up reactions without being consumed. Graphical methods of measuring reaction rates include gas collection, mass loss, and colour change. The section on reversible reactions explains equilibrium in closed systems and how changes in temperature, pressure, or concentration shift equilibrium, guided by Le Châtelier’s principle. Examples include hydrated copper sulfate and cobalt chloride equilibrium systems. The document then details two major industrial processes: the Haber process for ammonia production and the Contact process for sulfuric acid manufacture, explaining chosen conditions for yield and economic efficiency. Finally, redox reactions are introduced, defining oxidation as loss of electrons and reduction as gain, with rules for assigning oxidation numbers and identifying redox processes. This guide provides essential understanding of chemical reactivity and applications.
باز نشر محتواها در فضای مجازی، ممنوع است.
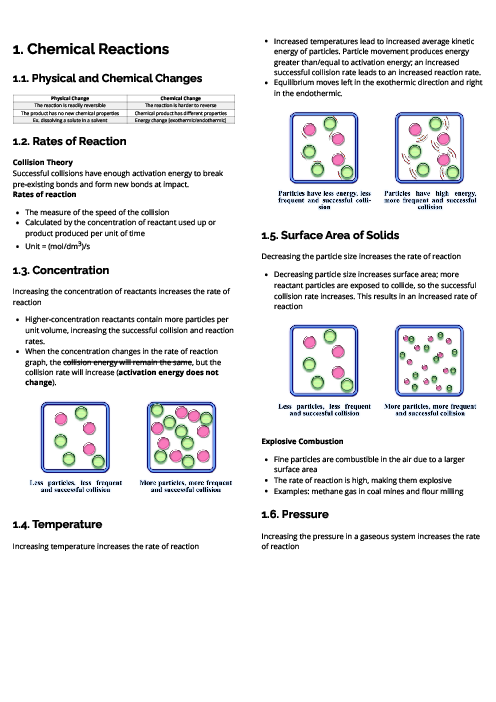
باز نشر محتواها در فضای مجازی، ممنوع است.