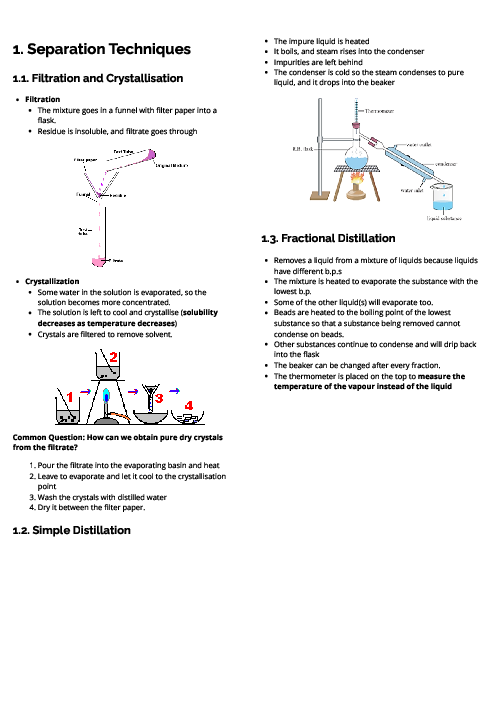
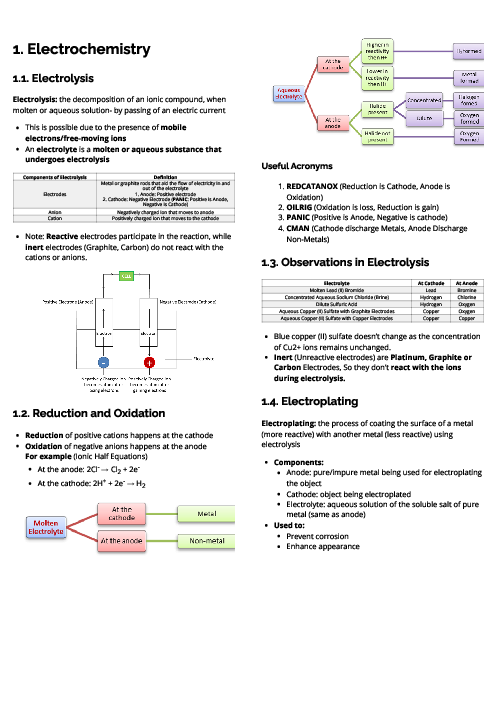
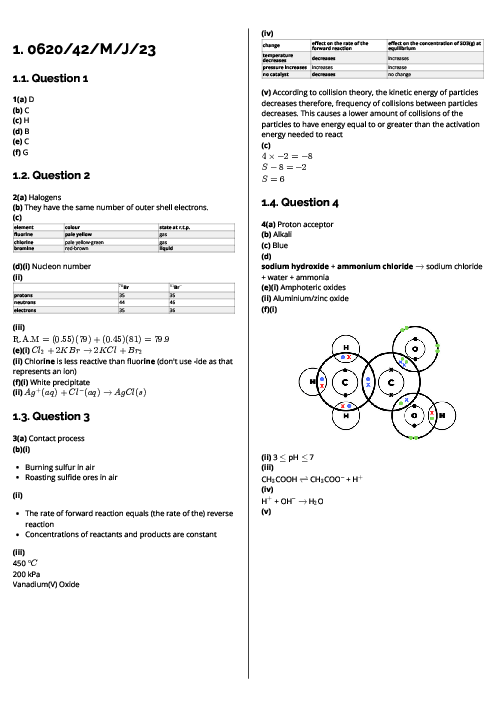
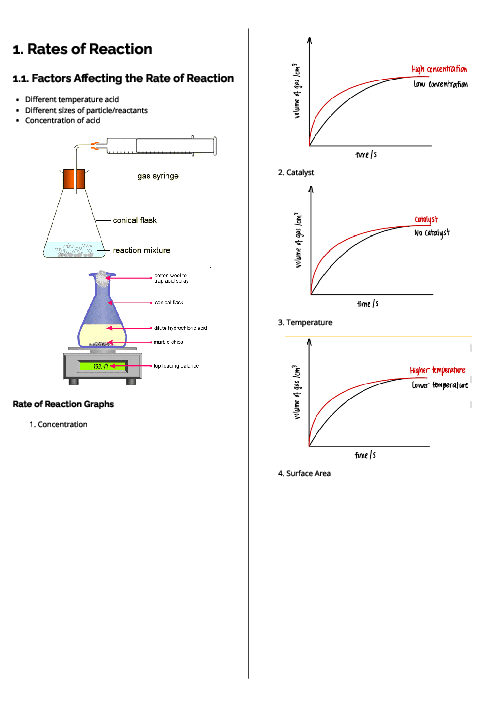
Chemistry (0620) Chemical Energetics Revision Note
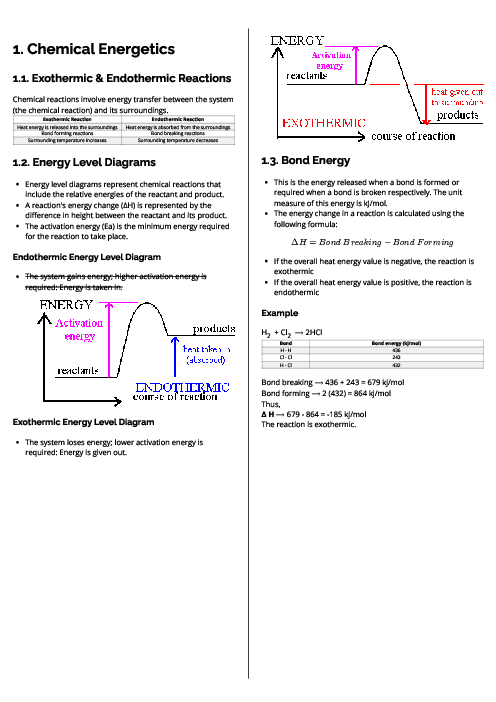
This document explains chemical energetics for CAIE IGCSE Chemistry, focusing on exothermic and endothermic reactions, energy level diagrams, and bond energies. It begins with the distinction between exothermic reactions, where heat is released into the surroundings and temperature rises, and endothermic reactions, where heat is absorbed and temperature falls. Energy level diagrams are introduced to illustrate the relative energies of reactants and products, with activation energy (Ea) shown as the minimum energy needed for a reaction to occur. Exothermic reactions are represented by a downward energy change (negative ΔH), while endothermic reactions show an upward energy change (positive ΔH). The concept of bond energy is explained as the energy required to break bonds or released when bonds form, measured in kJ/mol. The overall energy change in a reaction is calculated using the formula:
ΔH = Bond Breaking – Bond Forming.
Worked examples, such as the reaction between hydrogen and chlorine to form hydrogen chloride, demonstrate how to apply bond energy values to determine whether a reaction is exothermic or endothermic. This guide equips students with the essential understanding of energy transfers in reactions, preparing them to interpret energy changes, calculate enthalpy differences, and analyse reaction energetics.
باز نشر محتواها در فضای مجازی، ممنوع است.
باز نشر محتواها در فضای مجازی، ممنوع است.