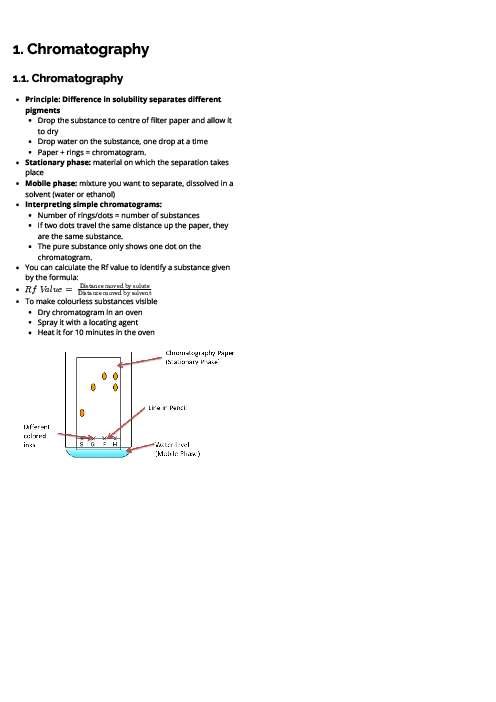
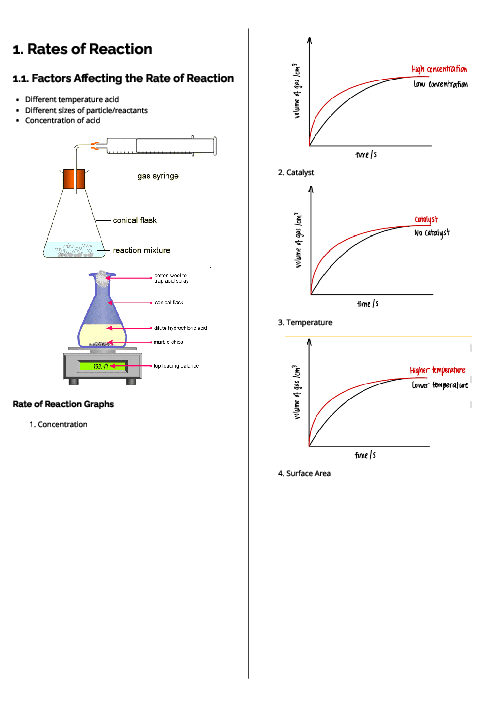
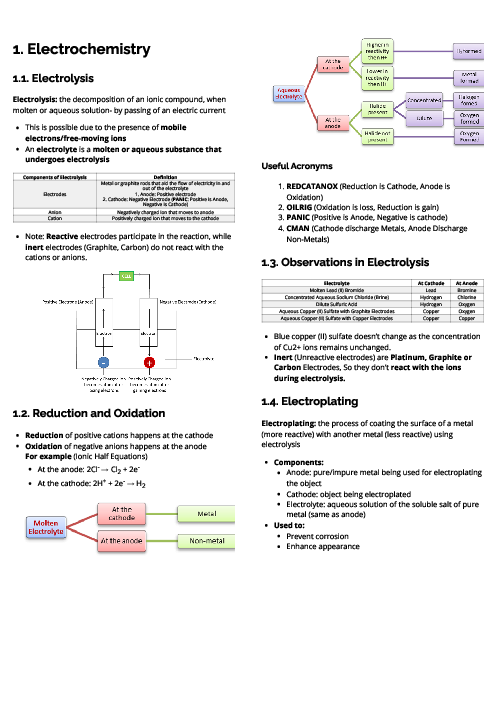
Chemistry (0620) Acids, Bases and Salts Revision Note
This document explains acids, bases, and salts for CAIE IGCSE Chemistry, focusing on their properties, reactions, and preparation methods. Acids are defined as proton donors that release hydrogen ions (H⁺) in water, turning blue litmus red and methyl orange red. Strong acids fully dissociate, while weak acids partially dissociate. Bases are proton acceptors that produce hydroxide ions (OH⁻), turning red litmus blue and methyl orange yellow; soluble bases are called alkalis. Strong alkalis completely dissociate, while weak alkalis only partially ionize. Neutral substances have pH 7, measured using universal indicators. The guide explains acid-base reactions, such as acids with metals (forming salt and hydrogen), bases (forming salt and water), or carbonates (forming salt, CO₂, and water). Types of oxides are outlined: basic (metal oxides), acidic (non-metal oxides), amphoteric (aluminium and zinc oxides), and neutral (CO and N₂O). A table of transition metal compound colours is included. The concept of water of crystallisation is explained in hydrated salts, along with general solubility rules for common compounds. Methods of salt preparation are described: using excess insoluble bases, titration for soluble salts, and precipitation for insoluble salts. This resource equips students with key chemical principles and practical techniques for exam success.
باز نشر محتواها در فضای مجازی، ممنوع است.
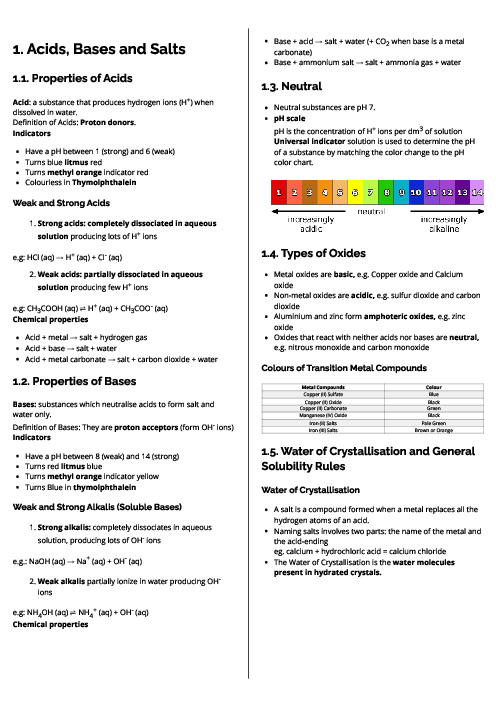
باز نشر محتواها در فضای مجازی، ممنوع است.